Two new publications highlight recent research from the UBC-based lab of NMIN researcher Dr. Sarah Hedtrich.
Transmucosal drug delivery
 The first, published 14 March 2021 in Small, describes the lab’s recent efforts to develop a method for efficient transmucosal drug delivery.
The first, published 14 March 2021 in Small, describes the lab’s recent efforts to develop a method for efficient transmucosal drug delivery.
“Mucosal surfaces pose a challenging environment for efficient drug delivery,” note the paper’s authors. To address this need, the researchers developed a synthesis of novel disulfide-containing dendritic polyglycerol (dPG)-based nanogels and subjected them to preclinical testing.
“Absorption studies in porcine intestinal tissue and human bronchial epithelial models demonstrate that disulfide-containing nanogels are highly efficient in overcoming mucosal barriers,” the researchers conclude.
“The nanogels efficiently degrade and deliver the anti-inflammatory biomacromolecule etanercept into epithelial tissues yielding local anti-inflammatory effects.”
The paper also describes problems encountered due to the paucity of appropriate test systems, noting the importance of taking a combined and multifaceted approach to the preclinical testing of mucosal drug-delivery systems.
The model dilemma in biomedical research
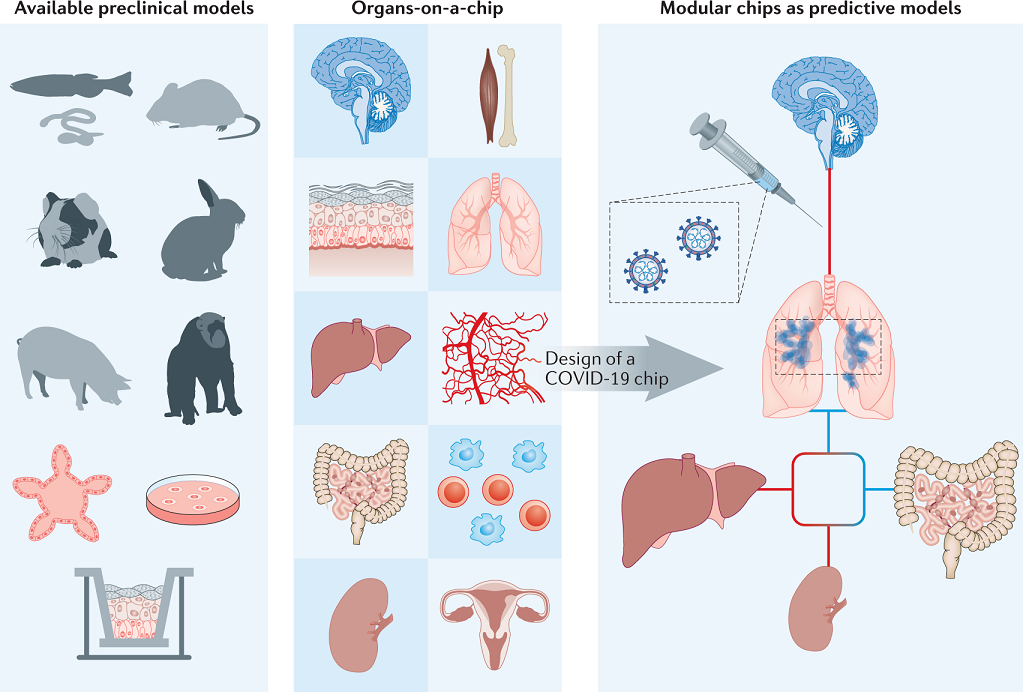
The second paper, published 17 March 2021 in Nature Reviews materials, considers how the biomedical response to COVID-19 reveals the need for new models for preclinical research.
“Scientists worldwide struggle to identify suitable animal models to study SARS-CoV-2 infections,” the researchers observe, noting that a World Health Organization international panel recently found that no animal model tested thus far entirely reflects human COVID-19.
More generally, “most animal models are poorly predictive of human conditions… [and] human-based models do not (yet) emulate complex human (patho)physiological processes and, thus, cannot simply replace animal models.”
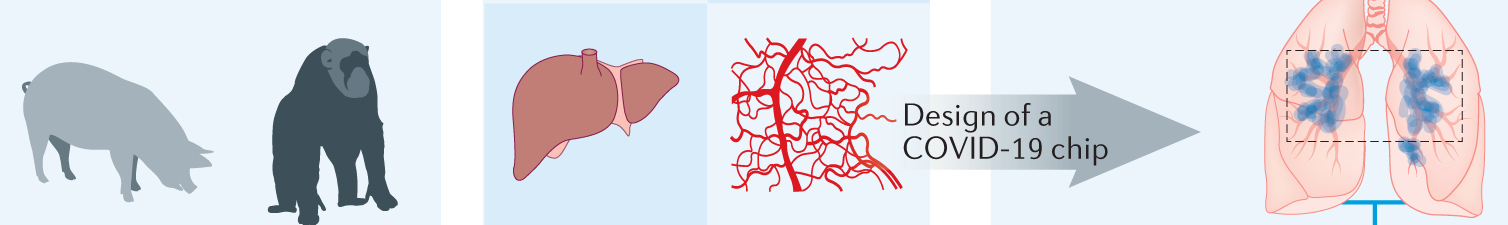
After assessing various approaches—including monolayer cultures, singularized 3D models, organs-on-a-chip, and humans-on-a-chip—the researchers suggest that “human-based models, such as micro-engineered multi-organs-on-chip, may hold the solution.” They further propose the design of customizable, ready-to-use modular chips using more optimal materials, and they also recommend that, for publications, “the reflexive request of data verification in animal models should be reassessed” as this need makes scientists reluctant to replace animal models with human-based cell and organ models.
Figure from Nature Reviews materials article: Modular ready-to-use chips as human (patho)physiology models.
The Hedtrich Lab combines research expertise in pharmacology, biomedical engineering and drug delivery and focuses on the establishment of next generation therapies, nanomedicine, and bioengineering. Dr. Sarah Hedtrich, PhD, is an Assistant Professor in the UBC Faculty of Pharmaceutical Sciences and is a co-investigator or collaborator on four NMIN-supported research projects in Themes I: Targeted Drug Delivery and II: Gene Therapy.